
Industries
Your role, your challenges ⭐

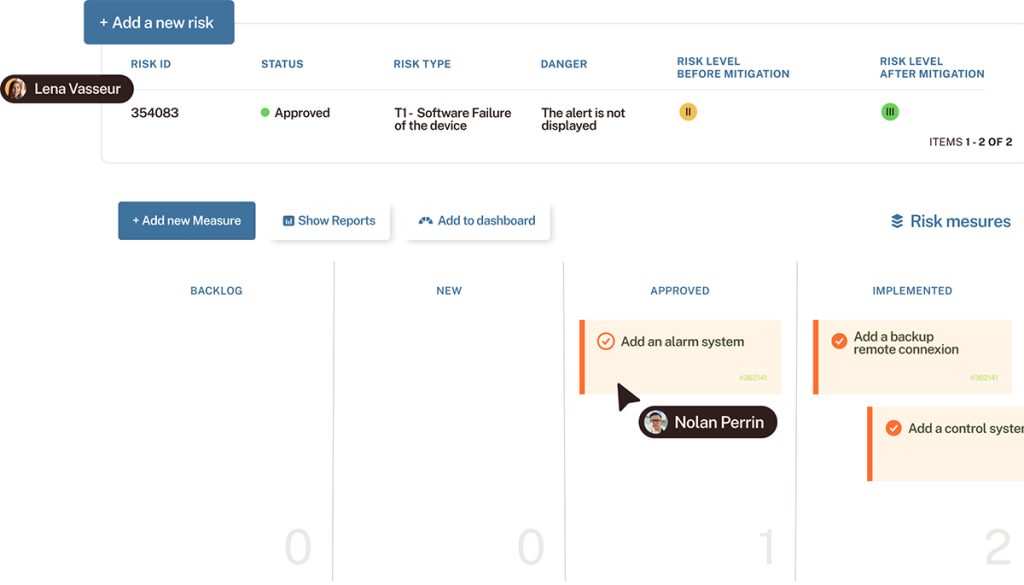
Discover Tuleap
Centralize requirements, traceability, tests, and documentation to ensure compliance and speed up delivery, from prototype to certified system.
Features
Resources
Latest blog post
Requirements traceability
Latest replays
Latest ebook
Guide – Scrum deployment with Tuleap
Latest customer story

