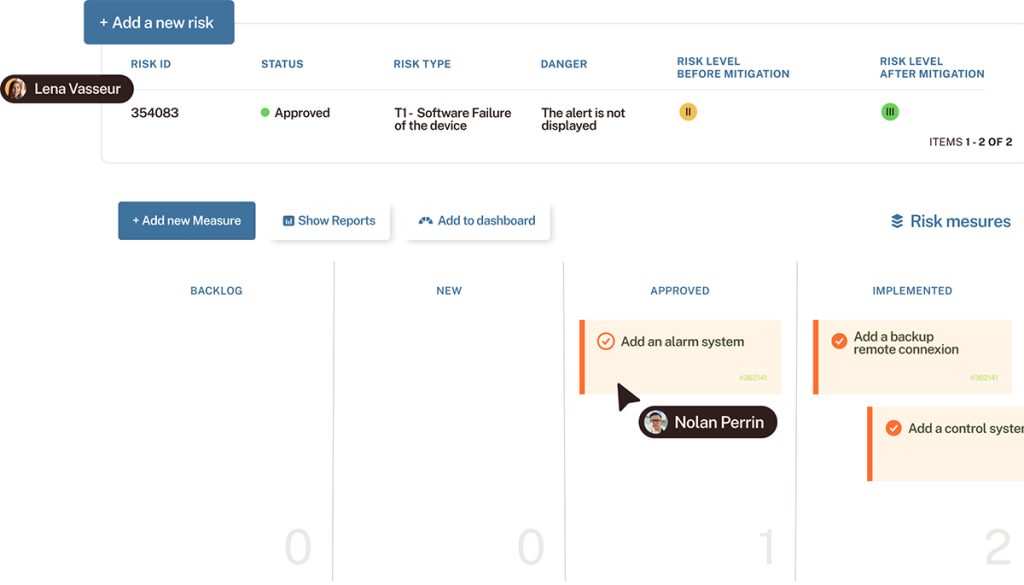
Program
- Structuring your requirements, tests, defects, and risks to ensure end-to-end traceability.
- Meeting IEC 62304 and ISO 13485 requirements using a single, unified tool.
- Aligning R&D, quality, and regulatory teams without creating silos.
- Preparing for audits with confidence (and without spreadsheets).
- A concrete example of a quality process supported by Tuleap.
- Real-world feedback: testimonials from medtech professionals (include the Softway story).